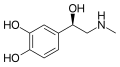
Epinephrine (also known as adrenaline, adrenalin, or β,3,4-trihydroxy-N-methylphenethylamine) is a hormone and a neurotransmitter. Epinephrine and norepinephrine are two separate but related hormones secreted by the medulla of the adrenal glands. They are also produced at the ends of sympathetic nerve fibres, where they serve as chemical mediators for conveying the nerve impulses to effector organs. The investigation of the pharmacology of epinephrine made a major contribution to the understanding of the autonomic system and the function of the sympathetic system. Epinephrine remains a useful medicine for several emergency indications. This is despite its non-specific action on adrenoceptors and the subsequent development of multiple selective medicines that target subtypes of the adrenoceptors. The word adrenaline is used in common parlance to denote increased activation of the sympathetic system associated with the energy and excitement of the fight-or-flight response. The influence of adrenaline is mainly limited to a metabolic effect and bronchodilation effect on organs devoid of direct sympathetic innervation.
In chemical terms, epinephrine is one of a group of monoamines called the catecholamines. It is produced in some neurons of the central nervous system, and in the chromaffin cells of the adrenal medulla from the amino acids phenylalanine and tyrosine.
Pathology
Increased epinephrine secretion is observed in phaeochromocytoma, hypoglycaemia, myocardial infarction and to a lesser degree in benign essential familial tremor. A general increase in sympathetic neural activity is usually accompanied by increased adrenaline secretion, but there is selectivity during hypoxia and hypoglycaemia, when the ratio of adrenaline to noradrenaline is considerably increased. Therefore, there must be some autonomy of the adrenal medulla from the rest of the sympathetic system.
Myocardial infarction is associated with high levels of circulating epinephrine and norepinephrine, particularly in cardiogenic shock.
Benign familial tremor (BFT) is responsive to peripheral β-adrenergic blockers and beta 2 stimulation is known to cause tremor. Patients with BFT were found to have increased plasma epinephrine, but not norepinephrine.
Low, or absent, concentrations of epinephrine can be seen in autonomic neuropathy or following adrenalectomy. Failure of the adrenal cortex, as with Addisons disease, can suppress epinephrine secretion as the activity of the synthesing enzyme, phenylethanolamine-N-methyltransferase, depends on the high concentration of cortisol that drains from the cortex to the medulla.
Medical uses
Adrenaline is used to treat a number of conditions including: cardiac arrest, anaphylaxis, and superficial bleeding. It has been used historically for bronchospasm and hypoglycemia, but newer treatments for these that are selective for beta2 adrenoceptors, such as salbutamol, a synthetic epinephrine derivative, are currently preferred. Currently the maximum recommended daily dosage for patients in a dental setting requiring local anesthesia with a peripheral vasoconstrictor is 10Â mcg/lb of total body weight
Cardiac arrest
Adrenaline is used as a drug to treat cardiac arrest and other cardiac dysrhythmias resulting in diminished or absent cardiac output. Its actions are to increase peripheral resistance via α1receptor-dependent vasoconstriction and to increase cardiac output via its binding to β1 receptors. The goal of reducing peripheral circulation is to increase coronary and cerebral perfusion pressures and therefore increase oxygen exchange at the cellular level. While epinephrine does increase aortic, cerebral, and carotid circulation pressure, it lowers carotid blood flow and end-tidal CO2 or ETCO2 levels. It appears that epinephrine may be improving macrocirculation at the expense of the capillary beds where actual perfusion is taking place. ETCO2 levels have become the marker that predicts the effectiveness of CPR and return of spontaneous circulation. The ability of epinephrine to increase macrocirculatory pressures does not necessarily increase blood flow through end organs. ETCO2 levels might more accurately reflect tissue perfusion instead of perfusion pressure markers.
Epinephrine has not demonstrated its ability to improve tissue perfusion or positively impact long term survival, and could be reducing the survival rates of patients in cardiac arrest.
Anaphylaxis
Epinephrine/adrenaline is the drug of choice for treating anaphylaxis. Allergy patients undergoing immunotherapy may receive an adrenaline rinse before the allergen extract is administered, thus reducing the immune response to the administered allergen.
Different strengths, doses and routes of administration of epinephrine are used for several medical emergencies. The commonly used Epipen Auto-Injector delivers 0.3Â mg epinephrine injection (0.3 mL, 1:1000) is indicated in the emergency treatment of allergic reactions (Type I) including anaphylaxis to stings, contrast agents, medicines or patients with a history of anaphylactic reactions to known triggers. A single dose is recommended for patients who weigh 30Â kg or more, repeated if necessary. A lower strength product is available for paediatric use This dose causes vasoconstriction at the site of subcutaneous injection, delaying absorption. The pharmacokinetic profile produces plasma concentrations in the region of 2 nmol/l, a concentration that be also be achieved with the repeated use of an epinephrine inhaler. This concentration is similar to that achieved during intense exercise and is too low to have significant beta 1 adrenoceptor, or alpha vasoconstrictor activity, but has marked beta 2 adrenceptor activity causing a fall in plasma potassium, an elevation in plasma glucose and enhanced finger tremor with additional bronchodilation and bronchoprotection.
The allergic reaction dose of epinephrine (0.1ml/kg 1/1000 Epinephrine with a max single dose of 0.3ml SQ or IM with IM being preferred in a low perfusion state) suppresses the experimentally induced wheal of the wheal and flare response to intradermally injected antigen. This suppression of the wheal is mediated by beta 2 adrenoceptors. The mechanism of the suppression of the edema is to reduce the vascular leak of fluid through the inter-endothelial junction of post capillary venules via beta receptor stimulation on the endothelial surface. Repeated doses, or higher doses, may cause additional microvascular constriction via alpha receptor stimulation, which also suppresses inflammatory edema.
When given intravenously (iv), instraosseous (IO) or intra-muscularly (im), the potency of epinephrine is greatly enhanced. For this reason for refractory anaphylactic shock or cardiac arrest a dilute epinephrine preparation is used of 1/10,000, the iv or im route being used for faster onset of action. The adult dose for refractory anaphylactic shock is usually 1 mg (1:10,000) IV/IO over 5 min and for cardiac arrest 1 mg (1:10,000) as an IV/IO push. When given IV/IO a major mechanism is via alpha adrenoceptor mediated vasoconstriction, increasing central blood pressure, making more selective alpha adrenoceptor agonists an alternative treatment Intramuscular injection can be complicated in that the depth of subcutaneous fat varies and may result in subcutaneous injection, or may be injected intravenously in error, or the wrong strength used. Intramuscular injection does give a faster and higher pharmacokinetic profile when compared to SC injection
Because of various expressions of α1 or β2 receptors, depending on the patient, administration of adrenaline may raise or lower blood pressure, depending on whether or not the net increase or decrease in peripheral resistance can balance the positive inotropic and chronotropic effects of adrenaline on the heart, effects that increase the contractility and rate, respectively, of the heart.
The usual concentration for SC or IM injection is 0.15- 0.3Â ml of 1:1,000. It is available in the trade as epipen
Asthma
Adrenaline is also used as a bronchodilator for asthma if specific β2 agonists are unavailable or ineffective.
When given by the subcutaneous or intramuscular routes for asthma, an appropriate dose is 300-500 mcg.
Croup
Racemic epinephrine has historically been used for the treatment of croup. Racemic adrenaline is a 1:1 mixture of the dextrorotatory (d) and levorotatory (l) isomers of adrenaline. The l- form is the active component. Racemic adrenaline works by stimulation of the α-adrenergic receptors in the airway, with resultant mucosal vasoconstriction and decreased subglottic edema, and by stimulation of the β-adrenergic receptors, with resultant relaxation of the bronchial smooth muscle.
Local anesthetics
Adrenaline is added to injectable forms of a number of local anesthetics, such as bupivacaine and lidocaine, as a vasoconstrictor to slow the absorption and, therefore, prolong the action of the anesthetic agent. Due to epinephrine's vasoconstricting abilities, the use of epinephrine in localized anesthetics also helps to diminish the total blood loss the patient sustains during minor surgical procedures. Some of the adverse effects of local anesthetic use, such as apprehension, tachycardia, and tremor, may be caused by adrenaline. Epinephrine/adrenalin is frequently combined with dental and spinal anesthetics and can cause panic attacks in susceptible patients at a time when they may be unable to move or speak due to twilight anesthesia.
Autoinjectors
Adrenaline is available in an autoinjector delivery system. Auvi-Qs, Jexts, EpiPens, Emerade, Anapens, and Twinjects all use adrenaline as their active ingredient. Twinject, which is now discontinued, contained a second dose of adrenaline in a separate syringe and needle delivery system contained within the body of the autoinjector. Though both EpiPen and Twinject are trademark names, common usage of the terms is drifting toward the generic context of any adrenaline autoinjector.
Adverse effects
Adverse reactions to adrenaline include palpitations, tachycardia, arrhythmia, anxiety, panic attack, headache, tremor, hypertension, and acute pulmonary edema.
Use is contraindicated in people on nonselective β-blockers, because severe hypertension and even cerebral hemorrhage may result. Although commonly believed that administration of adrenaline may cause heart failure by constricting coronary arteries, this is not the case. Coronary arteries have only β2 receptors, which cause vasodilation in the presence of adrenaline. Even so, administering high-dose adrenaline has not been definitively proven to improve survival or neurologic outcomes in adult victims of cardiac arrest.
Terminology

Epinephrine is the hormone's United States Adopted Name and International Nonproprietary Name, though the more generic name adrenaline is frequently used. The term Epinephrine was coined by the pharmacologist John Abel (from the Greek for "on top of the kidneys"), who used the name to describe the extracts he prepared from the adrenal glands as early as 1897. In 1901, Jokichi Takamine patented a purified adrenal extract, and called it "adrenalin" (from the Latin for "on top of the kidneys"), which was trademarked by Parke, Davis & Co in the U.S. In the belief that Abel's extract was the same as Takamine's, a belief since disputed, epinephrine became the generic name in the U.S. The British Approved Name and European Pharmacopoeia term for this chemical is adrenaline and is indeed now one of the few differences between the INN and BAN systems of names.
Among American health professionals and scientists, the term epinephrine is used over adrenaline. However, pharmaceuticals that mimic the effects of epinephrine are often called adrenergics, and receptors for epinephrine are called adrenergic receptors or adrenoceptors.
Mechanism of action
As a hormone and neurotransmitter, epinephrine acts on nearly all body tissues. Its actions vary by tissue type and tissue expression of adrenergic receptors. For example, high levels of epinephrine causes smooth muscle relaxation in the airways but causes contraction of the smooth muscle that lines most arterioles.
Epinephrine acts by binding to a variety of adrenergic receptors. Epinephrine is a nonselective agonist of all adrenergic receptors, including the major subtypes α1, α2, β1, β2, and β3. Epinephrine's binding to these receptors triggers a number of metabolic changes. Binding to α-adrenergic receptors inhibits insulin secretion by the pancreas, stimulates glycogenolysis in the liver and muscle, and stimulates glycolysis in muscle. β-Adrenergic receptor binding triggers glucagon secretion in the pancreas, increased adrenocorticotropic hormone (ACTH) secretion by the pituitary gland, and increased lipolysis by adipose tissue. Together, these effects lead to increased blood glucose and fatty acids, providing substrates for energy production within cells throughout the body.
Measurement in biological fluids
Epinephrine may be quantified in blood, plasma, or serum as a diagnostic aid, to monitor therapeutic administration, or to identify the causative agent in a potential poisoning victim. Endogenous plasma epinephrine concentrations in resting adults are normally less than 10Â ng/L, but may increase by 10-fold during exercise and by 50-fold or more during times of stress. Pheochromocytoma patients often have plasma adrenaline levels of 1000-10,000Â ng/L. Parenteral administration of epinephrine to acute-care cardiac patients can produce plasma concentrations of 10,000 to 100,000Â ng/L.
Biosynthesis and regulation
Epinephrine is synthesized in the medulla of the adrenal gland in an enzymatic pathway that converts the amino acid tyrosine into a series of intermediates and, ultimately, epinephrine. Tyrosine is first oxidized to L-DOPA, which is subsequently decarboxylated to give dopamine. Oxidation gives norepinephrine. The final step in epinephrine biosynthesis is the methylation of the primary amine of noradrenaline. This reaction is catalyzed by the enzyme phenylethanolamine N-methyltransferase (PNMT) which utilizes S-adenosylmethionine (SAMe) as the methyl donor. While PNMT is found primarily in the cytosol of the endocrine cells of the adrenal medulla (also known as chromaffin cells), it has been detected at low levels in both the heart and brain.
Physiology
The adrenal medulla is a minor contributor to total circulating catecholamines, though it contributes over 90% of circulating epinephrine. Little epinephrine is found in other tissues, mostly in scattered chromaffin cells. Following adrenalectomy, epinephrine disappears below the detection limit in the blood stream.
The adrenals contribute about 7% of circulating norepinephrine, most of which is a spill over from neurotransmission with little activity as a hormone. Pharmacological doses of epinephrine stimulate α1, α2, β1, β2, and β3 adrenoceptors of the sympathetic nervous system. Sympathetic nerve receptors are classified as adrenergic, based on their responsiveness to adrenaline.
The term “adrenergic†is often misinterpreted in that the main sympathetic neurotransmitter is norepinephrine (noradrenaline), rather than epinephrine, as discovered by Ulf von Euler in 1946.
Epinephrine does have a β2 adrenoceptor mediated effect on metabolism and the airway, there being no direct neural connection from the sympathetic ganglia to the airway.
The concept of the adrenal medulla and the sympathetic nervous system being involved in the flight, fight and fright response was originally proposed by Cannon. But the adrenal medulla, in contrast to the adrenal cortex, is not required for survival. In adrenalectomized patients haemodynamic and metabolic responses to stimuli such as hypoglycaemia and exercise remain normal.
Epinephrine is important as a central neurotransmitter. In the periphery circulating epinephrine can stimulate the facilitatory noradrenaline pre-synaptic β receptor, though the significance of this is not clear. Beta blockade in man and adrenalectomy in animals show that endogenous epinephrine has significant metabolic effects.
Exercise
The main physiological stimulus to epinephrine secretion is exercise. This was first demonstrated using the dennervated pupil of a cat as an assay, later confirmed using a biological assay on urine samples. Biochemical methods for measuring catecholamines in plasma were published from 1950 onwards. Although much valuable work has been published using fluorimetric assays to measure total catecholamine concentrations, the method is too non-specific and insensitive to accurately determine the very small quantities of epinephrine in plasma. The development of extraction methods and enzyme-isotope derivate radio-enzymatic assays (REA) transformed the analysis down to a sensitivity of 1 pg for epinephrine. Early REA plasma assays indicated that epinephrine and total catecholamines rise late in exercise, mostly when anaerobic metabolism commences.
During exercise the epinephrine blood concentration rises partially from increased secretion from the adrenal medulla and partly from decreased metabolism because of reduced hepatic blood flow. Infusion of epinephrine to reproduce exercise circulating concentrations of epinephrine in subjects at rest has little haemodynamic effect, other than a small β2 mediated fall in diastolic blood pressure. Infusion of epinephrine well within the physiological range suppresses human airway hyper-reactivity sufficiently to antagonize the constrictor effects of inhaled histamine.
A link between what we now know as the sympathetic system and the lung was shown in 1887 when Grossman showed that stimulation of cardiac accelerator nerves reversed muscarine induced airway constriction. In elegant experiments in the dog, where the sympathetic chain was cut at the level of the diaphragm, Jackson showed that there was no direct sympathetic innervation to the lung, but that bronchoconstriction was reversed by release of epinephrine from the adrenal medulla. An increased incidence of asthma has not been reported for adrenalectomized patients; those with a predisposition to asthma will have some protection from airway hyper-reactivity from their corticosteroid replacement therapy. Exercise induces progressive airway dilation in normal subjects that correlates with work load and is not prevented by beta blockade. The progressive dilation of the airway with increasing exercise is mediated by a progressive reduction in resting vagal tone. Beta blockade with propranolol causes a rebound in airway resistance after exercise in normal subjects over the same time course as the bronchoconstriction seen with exercise induced asthma. The reduction in airway resistance during exercise reduces the work of breathing.
Psychology
Emotional response
Every emotional response has a behavioral component, an autonomic component, and a hormonal component. The hormonal component includes the release of epinephrine, an adrenomedullary response that occurs in response to stress and that is controlled by the sympathetic nervous system. The major emotion studied in relation to epinephrine is fear. In an experiment, subjects who were injected with epinephrine expressed more negative and fewer positive facial expressions to fear films compared to a control group. These subjects also reported a more intense fear from the films and greater mean intensity of negative memories than control subjects. The findings from this study demonstrate that there are learned associations between negative feelings and levels of epinephrine. Overall, the greater amount of epinephrine is positively correlated with an arousal state of negative feelings. These findings can be an effect in part that epinephrine elicits physiological sympathetic responses including an increased heart rate and knee shaking, which can be attributed to the feeling of fear regardless of the actual level of fear elicited from the video. Although studies have found a definite relation between epinephrine and fear, other emotions have not had such results. In the same study, subjects did not express a greater amusement to an amusement film nor greater anger to an anger film. Similar findings were also supported in a study that involved rodent subjects that either were able or unable to produce epinephrine. Findings support the idea that epinephrine does have a role in facilitating the encoding of emotionally arousing events, contributing to higher levels of arousal due to fear.
Memory
It has been found that adrenergic hormones, such as epinephrine, can produce retrograde enhancement of long-term memory in humans. The release of epinephrine due to emotionally stressful events, which is endogenous epinephrine, can modulate memory consolidation of the events, ensuring memory strength that is proportional to memory importance. Post-learning epinephrine activity also interacts with the degree of arousal associated with the initial coding. There is evidence that suggests epinephrine does have a role in long-term stress adaptation and emotional memory encoding specifically. Epinephrine may also play a role in elevating arousal and fear memory under particular pathological conditions including post-traumatic stress disorder. Overall, the general findings through most studies supports that “endogenous epinephrine released during learning modulate the formation of long-lasting memories for arousing eventsâ€. Studies have also found that recognition memory involving epinephrine depends on a mechanism that depends on B-adrenoceptors. Epinephrine does not readily cross the blood-brain barrier, so its effects on memory consolidation are at least partly initiated by B-adrenoceptors in the periphery. Studies have found that sotalol, a B-adrenoceptor antagonist that also does not readily enter the brain, blocks the enhancing effects of peripherally administered epinephrine on memory. These findings suggest that B-adrenoceptors are necessary for epinephrine to have an effect on memory consolidation.
For noradrenaline to be acted upon by PNMT in the cytosol, it must first be shipped out of granules of the chromaffin cells. This may occur via the catecholamine-H+ exchanger VMAT1. VMAT1 is also responsible for transporting newly synthesized adrenaline from the cytosol back into chromaffin granules in preparation for release.
In liver cells, adrenaline binds to the β-adrenergic receptor, which changes conformation and helps Gs, a G protein, exchange GDP to GTP. This trimeric G protein dissociates to Gs alpha and Gs beta/gamma subunits. Gs alpha binds to adenyl cyclase, thus converting ATP into cyclic AMP. Cyclic AMP binds to the regulatory subunit of protein kinase A: Protein kinase A phosphorylates phosphorylase kinase. Meanwhile, Gs beta/gamma binds to the calcium channel and allows calcium ions to enter the cytoplasm. Calcium ions bind to calmodulin proteins, a protein present in all eukaryotic cells, which then binds to phosphorylase kinase and finishes its activation. Phosphorylase kinase phosphorylates glycogen phosphorylase, which then phosphorylates glycogen and converts it to glucose-6-phosphate.
Regulation
The major physiologic triggers of adrenaline release center upon stresses, such as physical threat, excitement, noise, bright lights, and high ambient temperature. All of these stimuli are processed in the central nervous system.
Adrenocorticotropic hormone (ACTH) and the sympathetic nervous system stimulate the synthesis of adrenaline precursors by enhancing the activity of tyrosine hydroxylase and dopamine-β-hydroxylase, two key enzymes involved in catecholamine synthesis. ACTH also stimulates the adrenal cortex to release cortisol, which increases the expression of PNMT in chromaffin cells, enhancing adrenaline synthesis. This is most often done in response to stress. The sympathetic nervous system, acting via splanchnic nerves to the adrenal medulla, stimulates the release of adrenaline. Acetylcholine released by preganglionic sympathetic fibers of these nerves acts on nicotinic acetylcholine receptors, causing cell depolarization and an influx of calcium through voltage-gated calcium channels. Calcium triggers the exocytosis of chromaffin granules and, thus, the release of adrenaline (and noradrenaline) into the bloodstream.
Unlike many other hormones adrenaline (as with other catecholamines) does not exert negative feedback to down-regulate its own synthesis. Abnormally elevated levels of adrenaline can occur in a variety of conditions, such as surreptitious epinephrine administration, pheochromocytoma, and other tumors of the sympathetic ganglia.
Its action is terminated with reuptake into nerve terminal endings, some minute dilution, and metabolism by monoamine oxidase and catechol-O-methyl transferase.
History
Extracts of the adrenal gland were first obtained by Polish physiologist Napoleon Cybulski in 1895. These extracts, which he called nadnerczyna, contained adrenaline and other catecholamines. American ophthalmologist William H. Bates discovered adrenaline's usage for eye surgeries prior to April 20, 1896. Japanese chemist Jokichi Takamine and his assistant Keizo Uenaka independently discovered adrenaline in 1900. In 1901, Takamine successfully isolated and purified the hormone from the adrenal glands of sheep and oxen. Adrenaline was first synthesized in the laboratory by Friedrich Stolz and Henry Drysdale Dakin, independently, in 1904.
References
General references
External links
- U.S. National Library of Medicine: Drug Information Portal - Epinephrine


0 komentar :
Posting Komentar